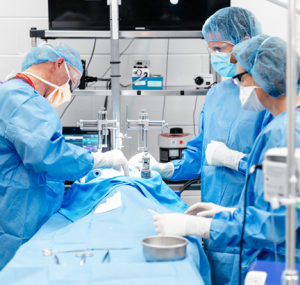
A Community of Scientists
We are not your typical GLP-compliant preclinical contract research organization (CRO); we are a collaborative community of scientists who believe in open communication, transparency, and strong partnerships. We partner with our sponsors, offering tailored solutions and expert guidance at every step. Our commitment to excellence, responsibility, and trust is unwavering, guiding us as we tackle some of the most challenging conditions in human health.

Consult a Scientist
We are a team of scientists who value open communication and collaboration. We work closely with our sponsors to provide customized solutions and expert support.
Our Expert Team
The Northern Bio Team is a group of highly motivated and talented people dedicated to execution, cutting-edge science, and advancing your therapeutic to the clinic. The team consists of scientists experienced in a range of therapeutic areas, accomplished operational leaders specialized in drug development, and compassionate veterinary staff experienced in the care of laboratory animals.
Michigan-Based Preclinical Research Facilities
With three research and development facilities located in Michigan, Northern Bio specializes in performing non-GLP and GLP-compliant research utilizing targeted drug delivery, molecular biology, and bioanalysis.
- Our Muskegon facility was designed for the execution of pre-clinical study in large animal species.
- Our Norton Shores facility is dedicated to high-quality rodent and ophthalmology research.
- Our Portage facility specializes in molecular biology and bioanalysis supporting preclinical and clinical trials.

Committed to Animal Welfare
At Northern Bio, we are devoted to the welfare of the laboratory animals in our care. We have the utmost compassion and respect for our animals. The extensive training that our staff receives supports the unwavering dedication to the principle of the 3Rs (replacement, reduction, and refinement). This refined approach safeguards the quality of life of the animals and ensures the success of your study.

Committed to Quality
Northern Bio’s independent quality assurance unit monitors all facility and GLP study activities. Our team integrates quality standards for GLP compliance (21 CFR Part 58) and electronic records compliance (21 CFR Part 11). Our team is made up of respected and experienced RQAP-GLP credentialed auditors providing regulatory oversite and guidance.
Partner With Us
Let’s work together to develop
life-changing treatments.
Join us in the pursuit of better lives through scientific innovation. Explore how Northern Bio can help you achieve your research goals.